This article was automatically translated from the original Turkish version.
+1 More
Significant figures are the digits in a measurement that determine its sensitivity and express the reliability of the measurement result. They are determined based on the precision of the measuring instrument and must be taken into account in calculations. Significant figures are of great importance in scientific calculations, engineering, physics and chemistry like.
Specific rules are followed to determine the significant figures of a number. These rules explain how the measurement result should be interpreted and how it should be handled during calculations.
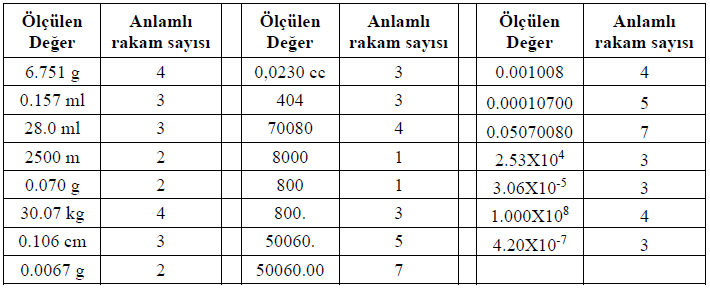
All non-zero digits are significant. For example:
4829 (This number has four significant figures.)
7.31 (It contains three significant figures.)
Zeros located between other digits are significant. For example:
505 (It contains three significant figures.)
30.06 (It contains four significant figures.)
Leading zeros are not significant. For example:
0.0045 (It contains two significant figures: 4 and 5.)
0.000306 (It contains three significant figures: 3, 0 and 6.)
If there is a decimal point: All zeros after the decimal point are significant.
10.00 (It contains four significant figures.)
0.500 (It contains three significant figures.)
If there is no decimal point: Trailing zeros may not be significant.
45000 (It contains two significant figures.)
3.200 × 10³ (It contains four significant figures; in scientific notation all digits are significant.)
Determining the Number of Significant Figures-KARADENİZ TECHNICAL UNIVERSITY
Exponential notation is a method used to write very large or very small numbers in a more short and understandable form. Numbers are written in the form a × 10ⁿ, where a is a decimal number between 1 and 10 and n is an complete integer.
The number 450000 is written in exponential notation as 4.5 × 10⁵ and contains two significant figures.
The number 0.0006789 is written in exponential notation as 6.789 × 10⁻⁴ and contains four significant figures.
The number 3.0200 × 10³ contains five significant figures because both the zeros and the decimal places are explicitly shown.
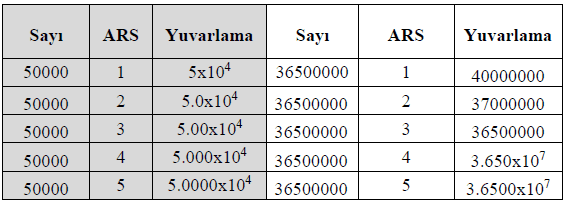
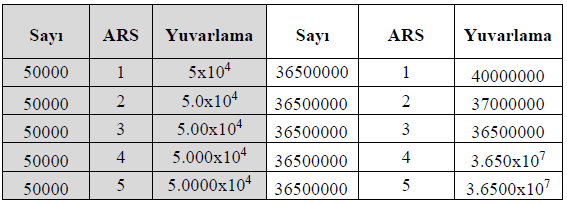
If a number must be expressed with a specific number of significant figures, rounding rules must be applied:
Expressing 4.56789 × 10⁵ with three significant figures: 4.57 × 10⁵.
Expressing 0.0034567 with two significant figures: 3.5 × 10⁻³.
Scientific notation allows numbers to be expressed more practically and better preserves the level of precision of measurements.
Exponential Notation-KARADENİZ TECHNICAL UNIVERSITY
In addition and subtraction, the result must have the same number of decimal places as the number with the least number of decimal places.
23.56 + 0.8 = 24.4 (The result matches the number with the least decimal places, 0.8.)
45.678 - 3.2 = 42.5 (Since 3.2 has the fewest decimal places, the result must be expressed with one decimal place.)
In multiplication and division, the result must have the same number of significant figures as the number with the least number of significant figures.
6.24 × 1.3 = 8.1 (Since 1.3 has two significant figures, the result must also have two significant figures.)
15.678 ÷ 2.1 = 7.5 (Since 2.1 has two significant figures, the result must also have two significant figures.)
Long calculation results must be rounded according to the rules for significant figures. When rounding, follow these rules:
If the digit to be dropped is 5 or greater, increase the preceding digit by one.
12.5678 → 12.57 (Rounded.)
6.789 → 6.79 (Rounded.)
If the digit to be dropped is 4 or less, the preceding digit remains unchanged.
12.344 → 12.34 (Rounded.)
9.8321 → 9.83 (Rounded.)
Rounding Significant Figures-KARADENİZ TECHNICAL UNIVERSITY
When evaluating measurement results, the concepts of accuracy and precision are distinct:
1. Precision: The repeatability of a measurement, meaning obtaining values close to each other. For example, measurements on a scale yielding values such as 1.21, 1.22, and 1.215 indicate high precision.
2. Accuracy: How close the measurement results are to the true value. For example, measuring a substance whose true value is 12.350 grams as 12.3495 grams indicates high accuracy.
A measurement can be precise but not accurate. For example, an object that actually weighs 9.85 grams may be measured as 10.00 grams. In this case, the measurement is precise but not accurate.
Significant figures are important in many scientific and technical fields:
1. Chemistry and Physics: Used to minimize measurement errors.
2. Engineering: Used in precise calculations and tolerance analysis.
3. Statistics: Used to ensure correct analysis of data.
4. Finance and Economics: Used in calculations requiring high precision.
No Discussion Added Yet
Start discussion for "Significant Figures" article
Determining Significant Figures
Non-Zero Digits
Zeros Between Digits
Leading Zeros
Trailing Zeros
Exponential Notation (Scientific Notation)
Significant Figures in Exponential Notation
Examples
Rounding in Exponential Notation
Arithmetic Operations with Significant Figures
Addition and Subtraction
Multiplication and Division
Rules for Rounding Numbers
Difference Between Accuracy and Precision
Applications of Significant Figures