This article was automatically translated from the original Turkish version.
+2 More

Yapay Zeka ile oluşturulmuştur.
Drug release refers to the process by which an active substance administered in a dosage form (e.g. tablet, capsule, implant) exits the form and exerts its effect under controlled or uncontrolled conditions. This process involves three fundamental steps: disintegration of the dosage form, disaggregation of particles, and dissolution.
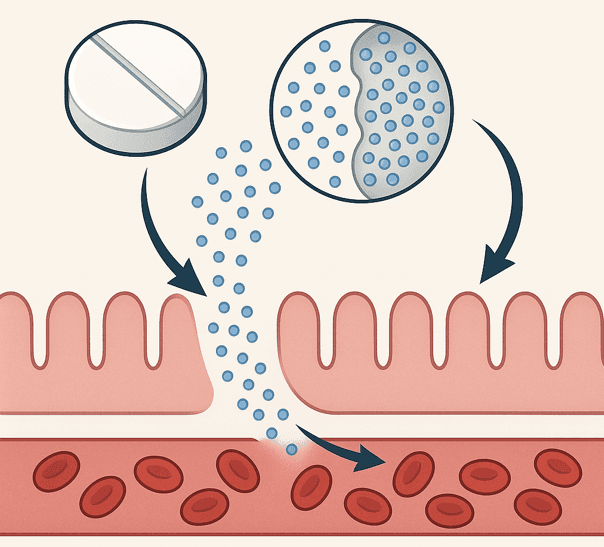
Controlled drug release (generated by Artificial Intelligence)
Controlled release systems aim to deliver the active substance at a predetermined rate and maintain constant plasma concentrations. They improve patient compliance by 40-85% and reduce the frequency of daily dosing by 50-75%.
Higuchi Equation (diffusion-based approach): The equation Q = KH · √t, used in diffusion-controlled matrix systems, calculates the cumulative amount of drug released over time. KH is the Higuchi constant, dependent on the drug’s diffusion coefficient, solubility, and matrix porosity. This model is valid for 20-80% release and shows a correlation coefficient R² > 0.95. Limitations of the Higuchi model include assumptions of a homogeneous matrix, the requirement of sink conditions, and complete dissolution. Due to these limitations, the Korsmeyer-Peppas model is preferred for complex release profiles.
Zero-Order Kinetics: Constant release rate independent of time, observed in osmotic and reservoir systems: dM/dt = k₀. This kinetic type is critical for drugs with a narrow therapeutic window (e.g. warfarin, digoxin).
Controlled release systems account for 37% of the total prescription drug market. Metformin XR formulations used in diabetes mellitus treatment have increased patient compliance by 73% and reduced gastrointestinal side effects by 45%.
Controlled release is essential for drugs with short half-lives (t½ < 4 hours). For example, while the half-life of propranolol is 3-6 hours, a controlled release formulation provides an effect duration of 12 hours.
Nanotechnology-based approaches include liposomes, nanocrystals, and polymeric nanoparticles. These systems are advancing targeted drug delivery and personalized medicine applications. Production costs are 200-400% higher than conventional systems.
Modern manufacturing technologies include 3D printing, electrospinning, and microencapsulation. These technologies enable the development of patient-specific dosage forms.
Drug release encompasses the entire process from disintegration of the dosage form to dissolution in the surrounding medium. Controlled release systems offer significant advantages in maintaining effective plasma levels, improving patient compliance, and reducing side effects. The release mechanism and type of polymer used directly influence the drug’s delivery profile.

Yapay Zeka ile oluşturulmuştur.
Controlled Release Systems
Main mechanisms
Kinetic Models
Polymers Used
Clinical Efficacy and Industrial Applications