This article was automatically translated from the original Turkish version.
+2 More
Electrochemical biosensors are a type of biosensor that converts biological or chemical changes into detectable signals for analysis. They are used to measure reference levels of numerous substances in the body, including glucose—the primary analyte in devices used by diabetic patients—as well as lactate, dopamine, CRP, cortisol, cholesterol, and toxins, thereby facilitating diagnosis. In clinical studies, they enable measurement of DNA and RNA sequences, viruses, bacteria, pathogens, and drug concentrations. Additionally, electrochemical biosensors are employed in food safety to detect highly harmful substances such as the mycotoxin aflatoxin B1, heavy metals Pb²⁺ (lead) and Hg²⁺ (mercury), and harmful agricultural pesticides.
Glucose Measurement Sensor (Pixabay)
In this type of biosensor, electrodes serve as transducers, converting biological or chemical events into measurable electrical signals such as current, voltage, or impedance. Electrochemical sensors based on redox reactions—chemical reactions involving electron transfer—generate a detectable signal through the electrodes. In redox terminology, a molecule that gains electrons is reduced, while one that loses electrons is oxidized. These electron transfer processes are known as redox couples. The oxidized and reduced species are described using the Nernst Equation, which forms the fundamental principle of operation for electrochemical biosensors.
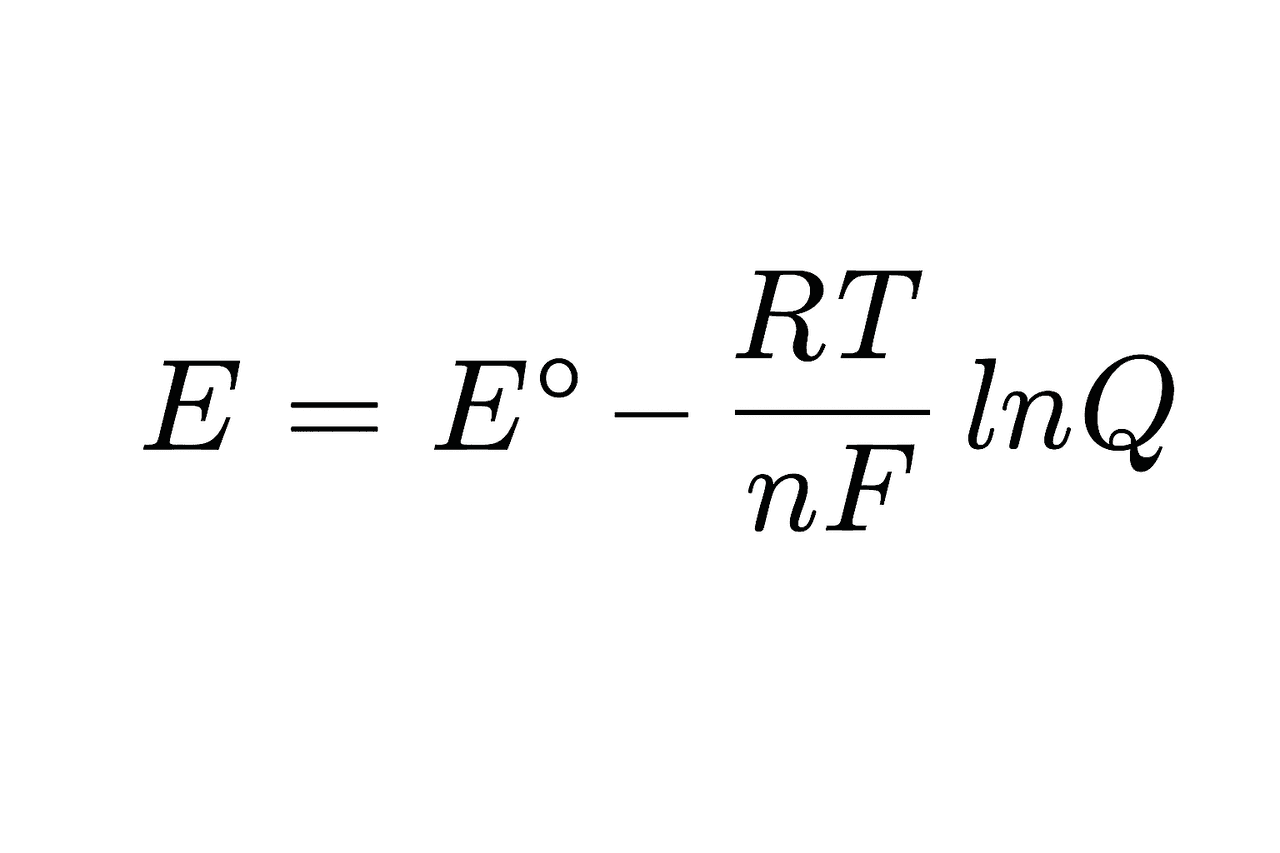
Nernst Equation (Generated by Artificial Intelligence)
The symbols in the equation are defined as follows:
E: Actual electrode potential
E∘: Standard electrode potential (1 M, 1 atm, 25°C)
R: Universal gas constant (8.314 J/mol·K)
T: Temperature (in Kelvin)
n: Number of electrons transferred in the reaction
F: Faraday constant (96485 C/mol e⁻)
Q: Ratio of reactant to product concentrations
As in other biosensors, the target substance being measured is called the analyte, and the molecule used to detect it is termed the biorecognition element. The analyte may consist of DNA or RNA sequences, proteins, antibodies, cells, or microorganisms.
Metal and carbon-based electrodes are commonly used. Carbon-based materials such as graphite and carbon fiber offer advantages due to their high chemical inertness and low electrical resistance. Other materials used in electrode fabrication include gold, silver, and stainless steel.
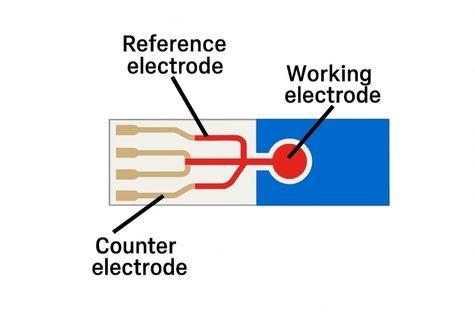
Electrode Structure (Generated by Artificial Intelligence)
In this setup, an insulating material is also present to prevent electrical short circuits between the electrodes. This insulator, made of materials such as PVC, PDMS, glass, or ceramic, enhances measurement control by facilitating the immobilization of biomolecules.
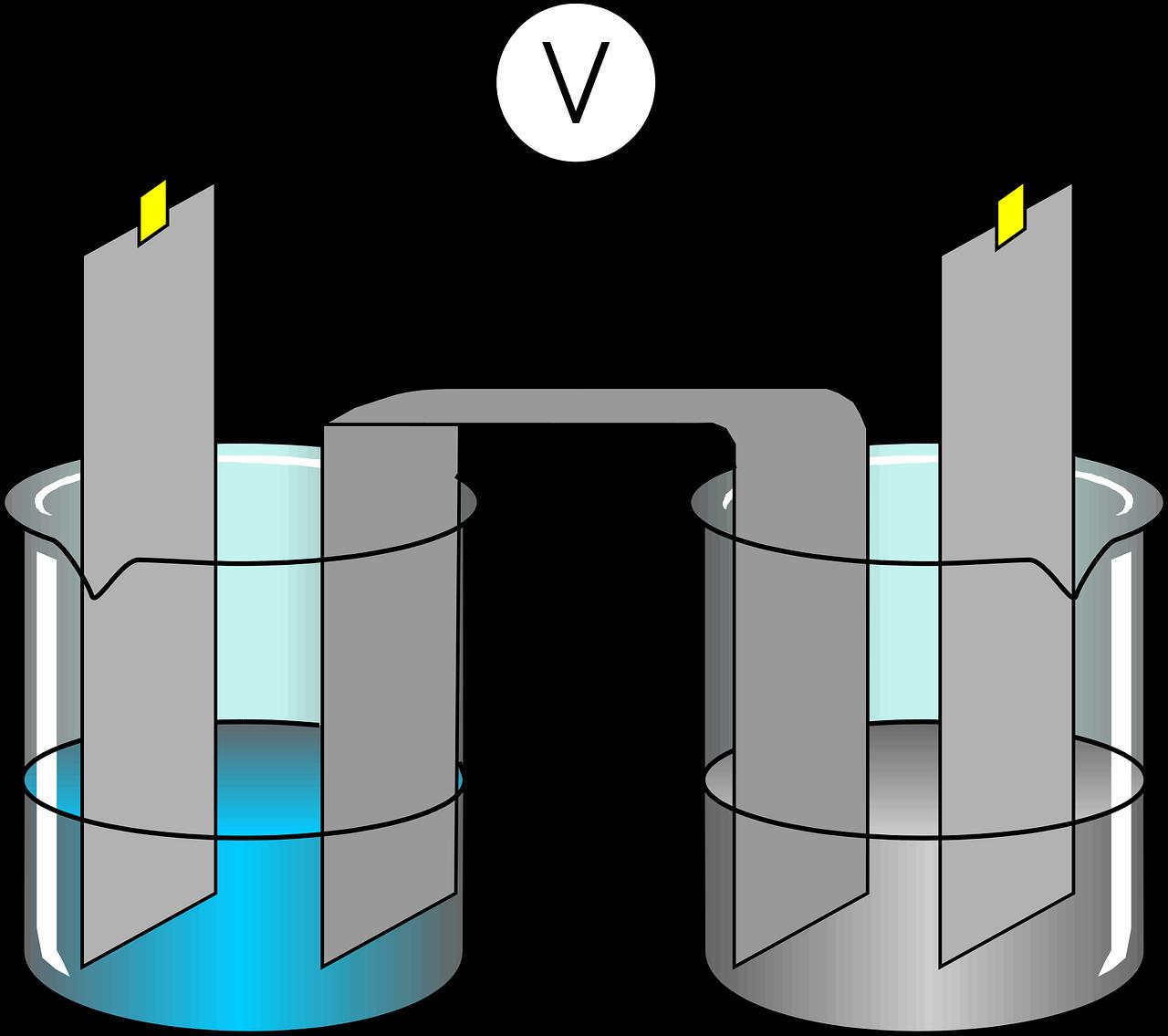
Electrochemical System Composed of Electrodes and Solutions (Pixabay)
Electrodes are immersed in an electrolyte solution that contains ions and enables electrical conduction. These solutions are typically composed of potassium ferrocyanide, phosphate-buffered saline (PBS), KCl, Na₂SO₄, HCl, NaOH, or saline water.
After redox reactions occur, changes in current and potential at the electrode surfaces are measured. The resulting signal, often extremely small at the micro- or nanoscale, is amplified. This analog signal is then converted into a digital format using an analog-to-digital converter for processing by computers and other digital systems. The processed signals are displayed as data on monitors or mobile screens for user interpretation.
Electrochemical biosensors used in point-of-care testing provide not only quantitative analysis but also high-sensitivity qualitative analysis. These features offer advantages over lateral flow assay (LFA) systems.
While certain drawbacks such as electrode fouling, the need for expert interpretation due to complex analyses, high calibration demands, and unwanted parasitic binding to electrodes are considered disadvantages compared to other point-of-care tests, electrochemical biosensors are valued for their ease of use, portability, minimal sample volume requirements, rapid results, and low cost.
The signal generated after a redox reaction is transmitted to the electrode through three main pathways:
These are methods used in electrochemical biosensors to identify analytes and determine their concentrations.
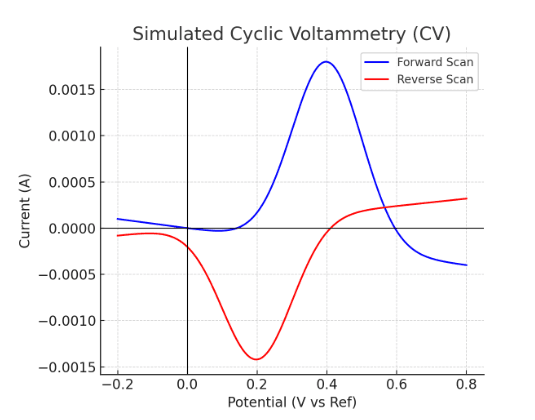
Cyclic Voltammetry Graph (Generated by Artificial Intelligence)
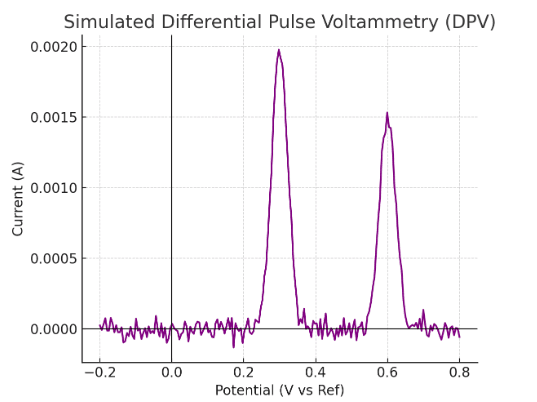
Differential Pulse Voltammetry Graph (Generated by Artificial Intelligence)
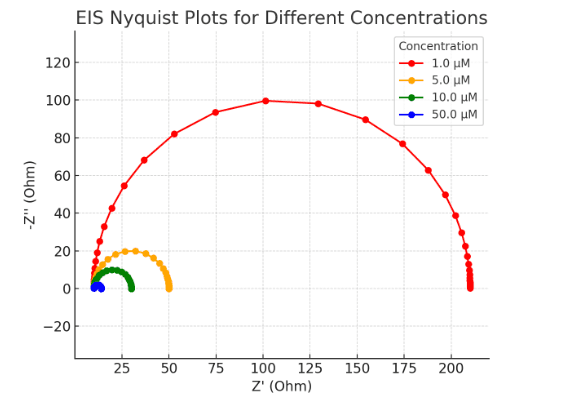
Electrochemical Impedance Spectroscopy Graph (Generated by Artificial Intelligence)
Clker-Free-Vector-Images. "Experiment-Electro-Chemical Laboratory" [Vektör grafik]. Pixabay. Accessed August 14, 2025.
LibreTexts. "Cyclic Voltammetry" [e-Kaynak]. Chemistry LibreTexts. Accessed August 14, 2025.
LibreTexts. "Optional Experiment – Differential Pulse Voltammetry" (DPV) [e-Laboratuvar kılavuzu]. Analysis of trace lead in water by anodic stripping voltammetry – Optional Experiment – Differential Pulse Voltammetry (DPV). Chemistry LibreTexts. Accessed August 14, 2025.
Sankar, K., Kuzmanović, U., Schaus, S. E., Galagan, J. E., and Grinstaff, M. W. "Strategy, Design, and Fabrication of Electrochemical Biosensors: A Tutorial." *ACS Sensors*, 9(5) (2024): 2254–2274. https://doi.org/10.1021/acssensors.4c00043
TesaPhotography. (2015, May 22). "Diabetes Finger Glucose" [Photograph]. Pixabay. Accessed August 14, 2025. https://pixabay.com/tr/photos/diyabet-parmak-glikoz-%C5%9Feker-hastas%C4%B1-777002/
Zhang, S., Wright, G., and Yang, Y. "Materials and Techniques for Electrochemical biosensor design and construction." Biosensors and Bioelectronics, 15(5–6), (2000): 273–282.
No Discussion Added Yet
Start discussion for "Electrochemical Biosensor" article
Electrode Structure
Operating Principle
Signal Transduction Pathways
Voltammetric Analysis Methods