This article was automatically translated from the original Turkish version.
+1 More
The lateral flow assay (LFA), also known as lateral flow or side-flow testing, is a biosensor system that offers users numerous advantages. Its working principle is based on capillary action—the phenomenon in which a liquid moves through a narrow space due to adhesive forces exceeding cohesive forces. LFA systems, as examples of point of care tests, provide rapid, easy, low-cost, portable, and reliable results. LFA-based test kits are used for diagnosing various viruses such as COVID-19, pregnancy, RSV, and influenza; toxins like pesticides and aflatoxins; illicit substances such as drugs; and chronic conditions.

Examples of Lateral Flow Tests Developed for SARS-CoV-2 Virus (Pexels)
The sample introduced into the test kit from a healthy individual or patient for the purpose of detecting the target analyte is called the sample. The actual substance sought for detection within the sample is referred to as the analyte. Depending on the presence or absence of the analyte and the working principle of the test kit, the desired analysis is obtained in a user-friendly manner.
LFA systems generally consist of five main components: the sample pad, conjugate pad, membrane, absorption pad, and backing.
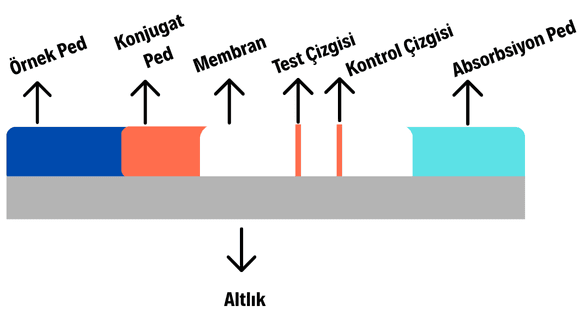
Visual Representation of the Internal Format of a Lateral Flow Test (Generated by Artificial Intelligence)
Sample Pad: This is the component through which the sample enters the test kit. Cellulose or glass fiber is typically used as the material. Cellulose allows for the production of a thicker and cheaper sample pad, while glass fiber provides better wicking ability, ensuring uniform delivery of the sample to the conjugate pad. Key parameters in designing the sample pad include bed volume (calculated by multiplying total volume and porosity), thickness, and particle retention rate. A higher bed volume increases sample absorption but slows down the flow rate within the test kit; this speed is adjusted according to the target test format and analyte. Additionally, a blocking solution is often applied via micropipette onto the sample pad to prevent binding of unwanted, non-specific substances present in the sample or environment and to regulate the sample’s pH. After drying under appropriate conditions, the treated pad is incorporated into the test kit.
Conjugate Pad: When the sample reaches the conjugate pad, it dissolves the detection antibodies present there, incorporating them into the capillary flow. Detection antibodies are labeled molecules such as silver or gold nanoparticles, enzymes, or fluorophores. The dissolution of these molecules upon sample arrival enables visible color development at the test and control lines, determining the test result. Materials such as cellulose, glass fiber, and polyester are commonly used in constructing the conjugate pad. Properly prepared detection antibodies require distinct preparation procedures (for example, silver and gold nanoparticles are precipitated via centrifugation before use). To ensure nanoparticle stability, proper dissolution upon sample arrival, and optimal flow performance, a blocking solution is either impregnated into or sprayed onto the conjugate pad, followed by drying under suitable conditions before integration into the test kit.
Membrane: The membrane, also known as the detection pad, contains the test and control lines. In LFA systems, the control line typically features a high-affinity capture secondary antibody, usually anti-IgG. The test line contains either a capture antibody or antigen, depending on the test format. These two lines are generally applied onto the membrane, typically made of nitrocellulose, either by direct drawing or dispensing.
Absorption Pad: This component absorbs excess fluid passing through the membrane, ensuring continuous capillary flow. Its size is determined based on the volume of liquid that must pass through the membrane.
Backing: Also referred to as the substrate, this component covers the entire bottom surface of the test kit and provides structural support. Polyvinyl alcohol (PVA) is commonly used for this purpose.
These components are cut to dimensions appropriate for the specific test format. In the final step, all components are assembled in the correct sequence on an adhesive backing. Typically, the developed test kit is first validated using pure water as a negative control. Subsequently, tests are performed using appropriate positive control substances. Then, specificity tests are conducted to determine whether the kit responds positively only to the target analyte and not to other substances. After all necessary validations are completed and the desired performance characteristics are confirmed, the test system composed of the pads and membrane is packaged appropriately. Even though it is not used in clinical laboratory settings, it provides a fast, reliable, and user-friendly diagnostic tool.
LFA systems are produced using two distinct mechanisms depending on the size of the analyte in the sample: the sandwich format and the competitive format.
In this format, the test line contains the antibody specific to the disease or virus being tested. The control line typically uses anti-IgG as a high-affinity capture antibody. The analyte, when present as an antigen, first binds to the detection antibody on the conjugate pad. The resulting antibody-antigen complex then binds to the capture antibody immobilized at the test line, positioning the analyte between two antibody molecules. This configuration is termed the sandwich format. Sandwich tests are used for detecting large molecules such as proteins and antigens; examples include tests for COVID-19, CRP, and HCG. In these tests, two lines indicate a positive result, while a single line indicates a negative result. The pregnancy test is the most iconic example of this format.
In this format, the test line contains the antigen specific to the disease or virus being tested. The control line, as in the sandwich format, typically uses anti-IgG, but a different antibody or molecule may be employed for control line formation depending on the test objective. The analyte in this case is the antibody specific to the target. The antibody introduced into the test kit competes with the labeled detection antibody (on the conjugate pad) for binding sites on the antigen immobilized at the test line—hence the name competitive format.
In the presence of the analyte, the labeled detection antibody (typically gold nanoparticles) is blocked from binding to the test line due to competition, resulting in no color development at the test line. Therefore, in the competitive format, a single line indicates a positive result. In the absence of the analyte, the detection antibody binds freely to the test line, producing a visible color. Thus, unlike the sandwich format, a double line appears when the analyte is absent (negative test), and a single line appears when the analyte is present (positive test). Competitive format LFA tests are preferred for analyzing small molecules such as drugs, toxins, hormones, illicit substances, and pesticides.

Example of a Lateral Flow Test (Pexels)
In LFA systems, the result is qualitative, determined by the presence of either one or two lines. Although they are weak in providing quantitative or numerical data, they offer significant advantages in terms of speed and practicality.
Both sandwich and competitive format lateral flow tests have become increasingly common today due to their long shelf life and user-friendly nature. Since the recent COVID-19 pandemic, there has been a substantial increase in demand for understanding and developing these tests. Many tests currently performed in specialized laboratory settings using more time-consuming methods such as ELISA and Western blot are now being adapted to LFA platforms.
No Discussion Added Yet
Start discussion for "Lateral Flow Assay (LFA)" article
Components
Working Principle
Sandwich Format
Competitive Format