This article was automatically translated from the original Turkish version.
Mixture is a collection formed by the combination of different types of atoms and molecules. substance substances consist either of a single element or a single compound, but most but substances are neither a single element nor a single compound. Many objects we encounter in daily life are not pure and belong to the category of mixtures. For example, the air we breathe, the food we eat chocolate, and the milk we drink are all examples of mixtures.
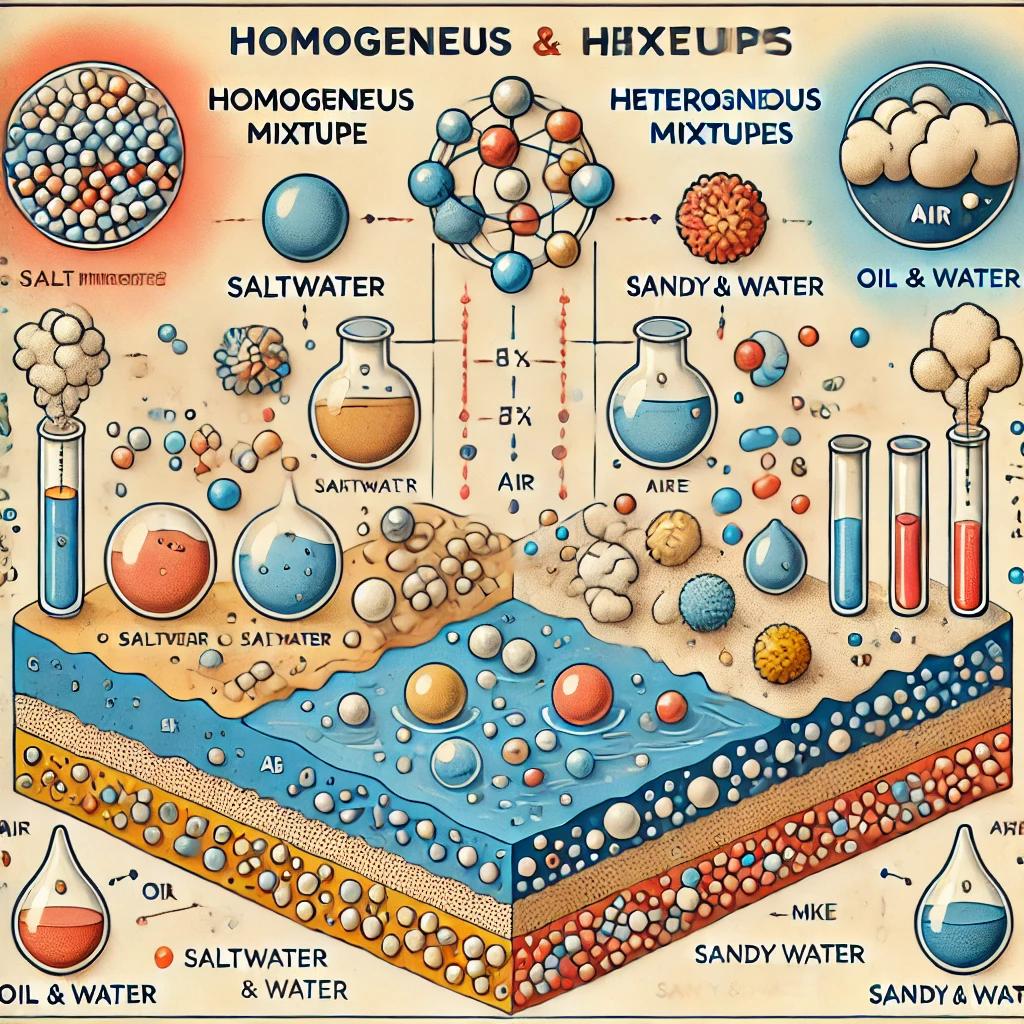
The components are evenly distributed throughout. They consist of a single phase. These types of mixtures are also called solutions. They can arise from solid-solid, liquid-gas, and gas-gas like interactions.

Solutions of substances under different physical conditions-Eokultv
Heterogeneous mixtures are those in which the composition is not uniform throughout, the components are not evenly distributed, and the substances do not dissolve in one another. They consist of multiple phases. Examples include sandy water, ink, fog, chalk-dust in water, and oil-in-water mixtures. When one substance in the mixture is dispersed within another, the dispersed substance is called the dispersed phase and the substance in which it is dispersed is called the dispersion phase. Heterogeneous mixtures are classified according to the physical states of the dispersion and dispersed phases:
Mixtures in which both the dispersion and dispersed phases are liquids are called emulsions. Examples include water-oil mixtures, water-diesel mixtures, milk, and mayonnaise.
Mixtures in which the dispersion phase is liquid and the dispersed phase is solid are called suspensions. Examples include chalk in iodine water, muddy water, sand-water, and ayran mixtures. In these mixtures, the solid phase settles over time due to the effect of duration place.
Heterogeneous mixtures in which the dispersion phase is gas and the dispersed phase is solid or liquid are called aerosols. If the dispersed phase is solid, it is called a solid aerosol; if it is liquid, it is called a liquid aerosol. Fog, spray, insect sprays, deodorants, and cloud are liquid aerosols. Smoke and dusty air are solid aerosols.
These are solid-solid heterogeneous mixtures. In ordinary (simple) mixtures, it is not possible to distinguish between the dispersed and dispersion phases. Examples include sand-sugar, salt-sugar, rice-bulgur, soil, and nut mixtures.
Colloids are mixtures in which a solid or liquid is suspended in a liquid or gas, appearing homogeneous but actually heterogeneous. This can be determined by observing that when a beam of light is passed through them, the light is scattered in all directions, or by microscopic examination, since homogeneous mixtures do not scatter light. The majority of emulsions and aerosols are also colloids. Examples include milk, smoke, mayonnaise, cream, jelly, soapy water, and blood.
Bikifi. "Heterojen Karışımlar." Accessed March 1, 2025. https://bikifi.com/biki/heterojen-karisimlar.
Bingöl Üniversitesi. "Genel Kimya Ders Sunumları." Accessed March 1, 2025. https://www.bingol.edu.tr/documents/Genel%20kimya%20Ders%20Sunumlar%C4%B1.pdf.
Bingöl Üniversitesi. "VI ve VII Hafta." Accessed March 1, 2025. https://www.bingol.edu.tr/documents/VI%20ve%20VII%20Hafta.pdf.
Eokul TV. "Karışımların Sınıflandırılması." 10. Sınıf. Accessed March 1, 2025. https://www.eokultv.com/karisimlarin-siniflandirilmasi-10-sinif/30628.

General Properties of Mixtures
Homogeneous Mixtures
Heterogeneous Mixtures
Emulsion
Suspension
Aerosol
Ordinary Mixture
Colloid