This article was automatically translated from the original Turkish version.
Stress corrosion cracking (SCC) is a specific type of cracking corrosion that occurs in metallic materials when a tensile stress (typically static or residual stress) and a corrosive environment are present simultaneously. During this process, microcracks develop without any visible corrosion signs on the metal surface, potentially leading to sudden and unexpected failures in the material. Indeed, unlike uniform (homogeneous) corrosion, SCC can cause the abrupt failure of critical components without any observable loss of material.
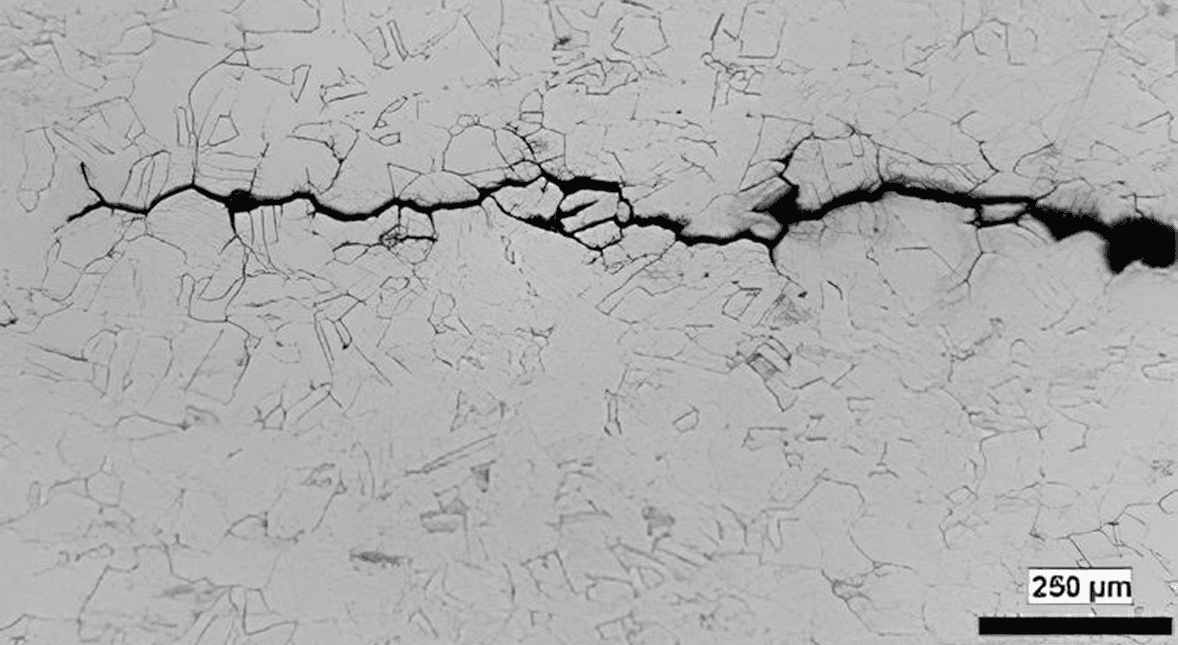
Stress Corrosion Cracking (Lou et al., 2017)
【1】
Cracks formed by SCC appear as thin, rust-filled fissures. When the crack propagates and the metal ultimately fractures, the fracture surface typically shows a portion heavily corroded and another portion clean but brittle. This indicates that the crack advanced slowly under corrosive influence, while the final rupture occurred suddenly due to mechanical failure. Stress corrosion cracks can initiate at stress levels far below the material’s yield strength. Thus, cracks may be triggered solely by corrosion effects under loads that the material would normally withstand. Moreover, these cracks are usually too small to be detected visually and often develop without any obvious surface corrosion products, making detection difficult. For this reason, SCC is recognized in industry as a damage mechanism that is hard to detect and can pose serious safety risks.
For stress corrosion cracking to occur, three essential factors must be present simultaneously: a suitable material structure, applied (or residual) tensile stress, and a supporting corrosive environment. The applied tensile stress on the material surface creates slip steps (dislocation movements) in the microstructure, leading to small fractures in the protective passive oxide layer. The corrosive environment penetrates through these fractured regions and causes localized anodic dissolution—that is, the selective dissolution of metal at the crack tip, weakening it. The stress concentration at the crack tip enables this localized corrosion to propagate the crack. As the crack grows, although stress intensity may temporarily decrease in that region, the reduction in cross-sectional area causes the average stress to rise again, accelerating crack propagation. This vicious cycle can lead to rapid crack growth and sudden fracture of the component in a short time.
SCC occurs only under tensile stresses. It does not occur under compressive stresses. Additionally, the nature of the stress is important. The described cracking typically occurs under constant tensile stresses. If the stress is variable or dynamic (repeated cyclic loading), the resulting damage combined with corrosion is termed corrosion fatigue and follows a different mechanism. Since cracks initiate at a microscopic scale, no visible surface changes occur initially. Over time, microcracks coalesce and grow, forming an internal crack network that remains invisible from the outside. When this network reaches a critical size, the material fails suddenly and in a brittle manner.

Intergranular and Transgranular Stress Corrosion Cracking (Generated by Artificial Intelligence)
Stress corrosion cracks can propagate with different morphologies depending on the material’s microstructure and the chemical environment. For instance, in some material-environment combinations, cracks follow grain boundaries (intergranular), while in others they traverse through the grains in an irregular path (transgranular). This behavior depends on the material’s internal structure (such as intergranular carbide precipitation, phase distribution, etc.) and the type of corrosion. For example, stainless steels typically exhibit transgranular SCC in chloride environments, but if the material has undergone sensitization such as intergranular chromium depletion, the cracks may follow grain boundaries. SCC is a highly complex damage mechanism requiring the simultaneous presence of mechanical stress and corrosion.
Each of the three factors—material, stress, and environment—plays a decisive role in SCC. First, the chemical composition and microstructure of the metal are critical. Stress corrosion cracking commonly occurs in specific alloys; alloys are generally more susceptible than pure metals. The material’s thermal history, grain structure, and the presence of second-phase particles or defects influence its susceptibility to SCC. For example, a high-carbon austenitic stainless steel (standard 304 grade) that has undergone intergranular carbide precipitation is more susceptible to SCC in chloride environments, whereas low-carbon or stabilized alloys (such as 316L stainless steel) exhibit significantly higher resistance. Similarly, duplex (two-phase) stainless steels, due to their balanced ferrite-austenite structure, demonstrate superior resistance to chloride-induced stress cracking and are therefore preferred in chemical processing and marine engineering applications.
Environmental conditions are a critical factor in SCC initiation. SCC often arises in the presence of specific chemical agents. For instance, chloride ions are the most common cause of SCC in stainless steels. Ammonia induces a type of SCC known as “season cracking” in copper-zinc alloys. Strong alkaline solutions can trigger SCC in carbon steels at elevated temperatures, known as “caustic embrittlement.” Strong acids or other specialized chemicals can also cause SCC in suitable materials. The concentration of dissolved oxygen in the environment also plays a role; in some alloys, the presence of oxygen disrupts the passive film and accelerates SCC.

Image of a Material Exhibiting Stress Corrosion Cracking (Popov, 2015)
【2】
Stress corrosion cracking occurs in metals subjected to tensile stress. This stress may arise not only from external loads but also from residual stresses resulting from welding, cooling after fabrication, or cold working. Consequently, welded or formed components are more prone to cracking in corrosive environments. Crack formation accelerates with increasing stress and temperature.
Stress corrosion cracking is one of the most critical damage mechanisms in industry and poses serious safety risks across multiple sectors. It is most commonly observed in the oil and gas, petrochemical, power generation (especially nuclear plants), marine, and aerospace industries. Equipment in these sectors typically consists of components subjected to high tensile stresses while simultaneously exposed to corrosive chemicals.

Cross-Sectional Image of a Fracture Caused by Stress Corrosion Cracking (Vandermaat et al., 2016)
【3】
For example, pipelines in petrochemical plants are susceptible to SCC due to internal pressure and chemical exposure. Stainless steel heat exchangers in nuclear reactors can develop cracks in hot, chloride-containing environments. In marine structures, continuous exposure to chloride ions in seawater under sustained stress leads to chloride-induced cracking. In aerospace, high-strength alloys used in landing gear, engine components, and fasteners may exhibit SCC over time in humid or salty environments.
These industries are particularly vulnerable to SCC because they operate under severe conditions involving high pressure, elevated temperatures, and aggressive chemicals. Moreover, commonly used materials such as stainless steels, nickel alloys, and titanium alloys are susceptible to cracking under inappropriate environmental conditions. For instance, sulfide-induced SCC is observed in refinery steels operating in sulfur-containing environments, while caustic SCC occurs in piping systems exposed to alkaline environments. Therefore, in these industries, early detection, regular monitoring, and careful material selection for SCC prevention are of critical importance.

Stress Corrosion Cracking in Threads of a Bolted Joint (Generated by Artificial Intelligence)
The most effective way to prevent stress corrosion cracking is to implement appropriate measures during material selection and design. First and foremost, materials chemically resistant to the operating environment should be selected. For example, replacing austenitic stainless steels with duplex stainless steels in chloride environments significantly reduces the risk of cracking.
Additionally, tensile stresses within the structure must be minimized. Residual stresses from welding or forming operations can be reduced through stress-relief annealing at appropriate temperatures. Furthermore, surface treatments that induce compressive stresses—such as shot peening—can prevent crack initiation.
Reducing the corrosive impact of the environment is another key strategy. This can be achieved by using corrosion inhibitors, controlling the concentration of harmful ions (particularly chlorides), and applying protective coatings.
Although SCC cannot always be entirely prevented, early detection is possible through regular inspections and non-destructive testing methods. Establishing a defined service life for critical equipment and replacing components before failure is also of great importance. When these measures are implemented together, the risk of SCC is substantially reduced and the safety of industrial facilities is significantly enhanced.
Féron, D., C. Guerre, E. Herms, and P. Laghoutaris. “9 – Stress Corrosion Cracking of Alloy 600: Overviews and Experimental Techniques.” In *Stress Corrosion Cracking of Nickel-Based Alloys in Water-Cooled Nuclear Reactors: The Coriou Effect*, 325–353. Cambridge: Woodhead Publishing, 2016. Accessed November 12, 2025. https://doi.org/10.1016/B978-0-08-100049-6.00009-4
Institute of Thermal Turbomachinery and Machine Dynamics, Graz University of Technology. “5.4.2.1 Fundamentals of Stress Corrosion Cracking.” *Aeroengine Safety*. Accessed November 12, 2025. https://aeroenginesafety.tugraz.at/doku.php?id=5%3A54%3A542%3A5421:5421
Lou, Xiaoyuan, Miao Song, Paul W. Emigh, Michelle A. Othon, and Peter L. Andresen. “On the Stress Corrosion Crack Growth Behaviour in High Temperature Water of 316L Stainless Steel Made by Laser Powder Bed Fusion Additive Manufacturing.” *Corrosion Science* 128 (2017): 140–153. Accessed November 12, 2025. https://doi.org/10.1016/j.corsci.2017.09.017
Popov, Branko N. “Chapter 1 – Evaluation of Corrosion.” In *Corrosion Engineering: Principles and Solved Problems*, 1–28. Amsterdam: Elsevier, 2015. Accessed November 12, 2025. https://doi.org/10.1016/B978-0-444-62722-3.00001-X
Vandermaat, D., S. Saydam, P. C. Hagan, and A. G. Crosky. “Examination of Rockbolt Stress Corrosion Cracking Utilising Full Size Rockbolts in a Controlled Mine Environment.” *International Journal of Rock Mechanics and Mining Sciences* 81 (2016): 86–95. Accessed November 12, 2025. https://doi.org/10.1016/j.ijrmms.2015.11.007
[1]
Xiaoyuan Lou et al., “On the Stress Corrosion Crack Growth Behaviour in High Temperature Water of 316L Stainless Steel Made by Laser Powder Bed Fusion Additive Manufacturing,” Corrosion Science 128 (2017): 140–53, https://doi.org/10.1016/j.corsci.2017.09.017
[2]
Branko N. Popov, “Chapter 1 – Evaluation of Corrosion,” in Corrosion Engineering: Principles and Solved Problems (Amsterdam: Elsevier, 2015), 1–28, https://doi.org/10.1016/B978-0-444-62722-3.00001-X
[3]
D. Vandermaat et al., “Examination of Rockbolt Stress Corrosion Cracking Utilising Full Size Rockbolts in a Controlled Mine Environment,” International Journal of Rock Mechanics and Mining Sciences 81 (2016): 86–95, https://doi.org/10.1016/j.ijrmms.2015.11.007
No Discussion Added Yet
Start discussion for "Stress Corrosion Cracking" article
Formation Mechanism
Influencing Factors
Industries Where SCC Is Observed
Prevention and Protection Methods