This article was automatically translated from the original Turkish version.
+1 More

Bisfenol A (BPA) (Yapay Zeka İle Oluşturulmuştur)
Bisphenol A (BPA) is a synthetic organic compound widely used in modern industry, serving as the primary monomer in the production of polycarbonate plastics and epoxy resins. With the chemical formula (CH3)2C(C6H4OH)2, BPA belongs to the class of diphenylmethane derivatives known as bisphenols, containing two hydroxyphenyl groups.
BPA was first synthesized in 1891 by Russian chemist Aleksandr P. Dianin. This synthesis was achieved through the condensation of phenol with acetone in the presence of an acid catalyst. The compound’s estrogenic (hormone-like) properties were discovered in the 1930s and demonstrated in vivo in 1936 by Dodds and Lawson. In the 1950s, the reaction of BPA with phosgene produced a hard, transparent resin known as polycarbonate, which rapidly expanded its industrial use. Today, BPA is among the highest-volume chemicals produced worldwide.
BPA is a solid substance with a molecular weight of 228.29 Da, appearing as white crystals or powder at room temperature. Its melting point is approximately 156oC. Structurally, it consists of two phenol molecules linked by an acetone molecule. Low vapor pressure and low volatility are key physical properties of BPA. It is lipophilic, with a water-octanol partition coefficient (logP) ranging from 3.32 to 3.4, indicating its potential to accumulate in fatty tissues. Its solubility in water is moderate, approximately 200 mg/dm3 at 25oC.
The most defining biological and toxicological feature of BPA is the striking structural similarity of its molecule to the body’s natural estrogen hormone, 17β-estradiol. This molecular mimicry is the fundamental reason for its function as an endocrine disruptor. Due to this similarity, BPA can bind to estrogen receptors (ERα and ERβ) in cells, thereby activating or blocking hormonal signaling pathways.
In industrial applications, BPA is the primary monomer responsible for the hard, transparent structure of polycarbonate plastics and epoxy resins. However, the chemical bonds formed during polymerization do not always remain stable. Factors such as heat, acidic or basic environments, and mechanical wear can cause hydrolysis of polymer chains, releasing free BPA molecules. Derivatives such as Bisphenol S (BPS) and Bisphenol F (BPF), produced by chemical modification of the BPA molecule, possess nearly identical structural backbones.
BPA has a broad range of applications in modern industry. The majority of produced BPA (up to 95%) is used in the manufacture of polycarbonate plastics and epoxy resin.

Applications of BPA (Generated by Artificial Intelligence)
Due to the widespread use of BPA in industry, the human body is exposed to bisphenol A through various routes. These exposure pathways are broadly categorized as dietary (ingestion), dermal contact, and inhalation.
Bisphenol A (BPA) is an agent classified among “endocrine-disrupting compounds” (EDCs). Its mechanism of action is based on mimicking, blocking, or altering the functioning of the body’s natural endocrine (hormonal) system. These interactions occur at molecular, cellular, and genetic levels.
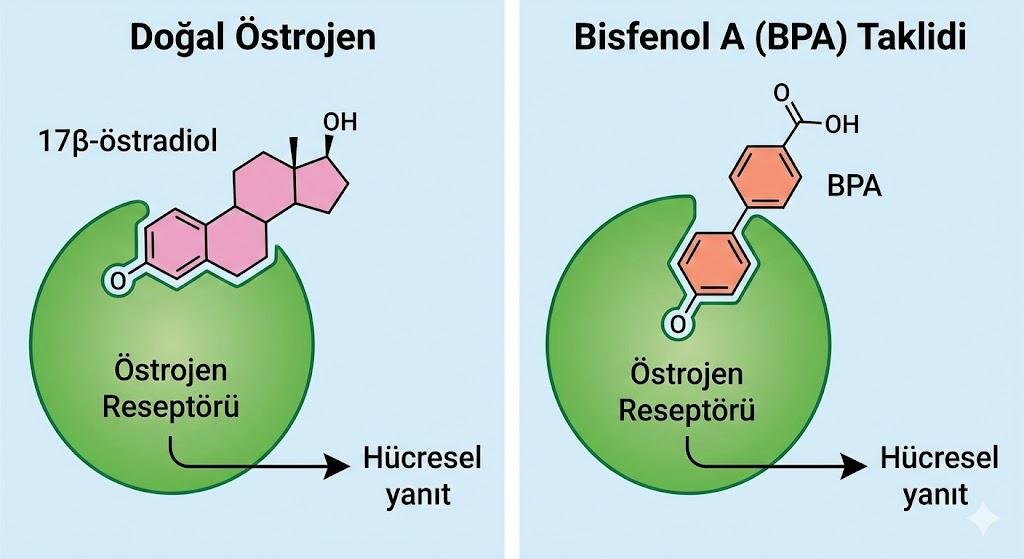
BPA (Right) Mimicking Natural Estrogen Hormone (Left) by Binding to Estrogen Receptor and Triggering Cellular Response (Lock-and-Key Model) (Generated by Artificial Intelligence)
Bisphenol A (BPA) exposure is considered an environmental risk factor contributing to the development of various chronic diseases due to its systemic effects. The most prominent health effects of BPA are linked to its obesogenic nature—that is, its ability to promote obesity. While traditional explanations for obesity pathogenesis focus on imbalances in caloric intake and energy expenditure, environmental chemicals are now recognized as playing a critical role. BPA directly affects adipose tissue biology by accelerating the transformation of preadipocytes into mature fat cells and increasing their lipid storage capacity. This biological mechanism is supported by epidemiological data, which show a positive correlation between high BPA exposure and increased body mass index (BMI) and waist circumference (abdominal obesity). This indicates that BPA can alter metabolic programming that controls body weight.
Another health issue closely associated with obesity is diabetes and glucose metabolism disorders. The metabolic effects of BPA are linked to disruption of glucose homeostasis and the development of type 2 diabetes. BPA impairs the function of insulin-secreting beta cells in the pancreas and induces insulin resistance in peripheral tissues. This mechanism weakens the body’s ability to regulate blood sugar, creating conditions for hyperglycemia. In the clinical picture of metabolic syndrome—characterized by the coexistence of obesity and insulin resistance—BPA exposure is identified as a triggering and accelerating factor.
In addition to metabolic effects, due to its estrogenic impact on the endocrine system, BPA also poses a potential threat to female reproductive physiology. By mimicking or blocking natural hormones, this chemical disrupts signaling in the hypothalamic-pituitary-ovarian axis, potentially causing menstrual irregularities and hormonal fluctuations. Its adverse effects on ovarian reserve, oocyte quality, and embryo development can reduce fertility potential. Literature also identifies BPA as an environmental contributor to the pathophysiology of reproductive disorders such as polycystic ovary syndrome (PCOS).
The environmental distribution of bisphenol A (BPA) is directly related to industrial production processes and the management of post-consumer waste. Release of the chemical into the environment occurs through wastewater from production facilities, leachate from landfills, and the physical or chemical degradation of plastics in nature. In regions with intensive industrial activity, such as China, significant levels of BPA have been detected in surface waters, river sediments, and wastewater treatment sludge. When wastewater treatment processes fail to fully remove BPA, discharge into receiving environments increases pollution loads in aquatic ecosystems.
In addition to water sources, BPA is also evaluated in terms of atmospheric transport and soil contamination. Due to its physical properties, BPA can adhere to airborne particles and be transported via dust, making it detectable in both indoor and outdoor air. Leachate from solid waste sites contaminating groundwater or the use of treatment sludge as agricultural fertilizer leads to soil pollution. This environmental cycle enables the chemical to move between water, air, and soil matrices, ensuring its persistence as a widespread contaminant across ecosystems.
Alternatives to bisphenol A used in the production of polycarbonate plastics and epoxy resins have become widespread in industrial applications. Products labeled “BPA-Free” or “Contains no BPA” typically use structural analogs of BPA, such as bisphenol S (BPS) and bisphenol F (BPF). These compounds exhibit significant structural similarity to BPA, preserving the physical durability, transparency, and heat resistance of the resulting plastics.
Toxicological studies and literature data indicate that BPS and BPF also exhibit biological effects similar to those of BPA. These alternative substances have been found to interfere with the endocrine system, displaying estrogenic and androgenic activities by interacting with hormonal receptors. Research on obesity and diabetes suggests that exposure to BPS and BPF is also associated with the development of these metabolic disorders. Their effects on adipose tissue biology and glucose metabolism confirm that these chemical substitutes possess comparable biological activity.
Warning: The content in this article is provided solely for general encyclopedic information purposes. The information herein should not be used for diagnosis, treatment, or medical advice. Before making any decisions regarding health, you must consult a physician or qualified healthcare professional. The author and KÜRE Encyclopedia assume no responsibility for any consequences arising from the use of this information for diagnosis or treatment purposes.
Alharbi, H. F., Raya Algonaiman, Rana Alduwayghiri et al. "Exposure to Bisphenol A Substitutes, Bisphenol S and Bisphenol F, and Its Association with Developing Obesity and Diabetes Mellitus: A Narrative Review." *International Journal of Environmental Research and Public Health* 19, no. 23 (2022): 15918. Accessed December 31, 2025. https://doi.org/10.3390/ijerph192315918.
Biemann, R., M. Blüher, and B. Isermann. "Exposure to Endocrine-Disrupting Compounds such as Phthalates and Bisphenol A is Associated with an Increased Risk for Obesity." *Best Practice & Research Clinical Endocrinology & Metabolism* 35, no. 5 (2021): 101546. Accessed December 31, 2025. https://doi.org/10.1016/j.beem.2021.101546.
Cimmino, Ilaria, Francesca Fiory, Giuseppe Perruolo et al. "Potential Mechanisms of Bisphenol A (BPA) Contributing to Human Disease." *International Journal of Molecular Sciences* 21, no. 16 (2020): 5761. Accessed December 31, 2025. https://doi.org/10.3390/ijms21165761.
García, Mónica García, Yolanda Picó, and María Morales-Suárez-Varela. "Effects of Bisphenol A on the Risk of Developing Obesity." *Nutrients* 16, no. 21 (2024): 3740. Accessed December 31, 2025. https://doi.org/10.3390/nu16213740.
Huang, Y. Q., C.K.C. Wong, J.S. Zheng, et al. "Bisphenol A (BPA) in China: A Review of Sources, Environmental Levels, and Potential Human Health Impacts." *Environment International* 42 (2012): 91-99. Accessed December 31, 2025. https://doi.org/10.1016/j.envint.2011.04.010.
Manzoor, Muhammad Faisal, Tayyaba Tariq, Birjees Fatima et al. "An Insight into Bisphenol A, Food Exposure and Its Adverse Effects on Health: A Review." *Frontiers in Nutrition* 9 (2022): 1047827. Accessed December 31, 2025. https://doi.org/10.3389/fnut.2022.1047827.
Panagopoulos, Periklis, Despina Mavrogianni, Chryssi Christodoulaki et al. "Effects of Endocrine Disrupting Compounds on Female Fertility." *Best Practice & Research Clinical Obstetrics and Gynaecology* 88 (2023): 102347. Accessed December 31, 2025. https://doi.org/10.1016/j.bpobgyn.2023.102347.
Pérez-Bermejo, Marcelino, Irene Mas-Pérez, and Maria Teresa Murillo-Llorente. "The Role of the Bisphenol A in Diabetes and Obesity." *Biomedicines* 9, no. 6 (2021): 666. Accessed December 31, 2025. https://doi.org/10.3390/biomedicines9060666.
Semerci, İbrahim Özay. "Bisfenol A: Azı Karar Çoğu Zarar mı?" TÜBİTAK Bilim ve Teknik Dergisi, (March 2014): 44–46. Accessed December 31, 2025. https://e-dergi.tubitak.gov.tr/edergi/yazi.pdf?dergiKodu=4&cilt=47&sayi=837&sayfa=44&yaziid=35952
Vandenberg, Laura N., Russ Hauser, Michele Marcus, et al. "Human Exposure to Bisphenol A (BPA)." *Reproductive Toxicology* 24, no. 2 (August-September 2007): 139-177. Accessed December 31, 2025. https://doi.org/10.1016/j.reprotox.2007.07.010.

Bisfenol A (BPA) (Yapay Zeka İle Oluşturulmuştur)
Chemical Structure and Properties
Applications
Exposure Pathways
Mechanism of Action
Health Effects
Environmental Impact
Regulations and Restrictions
BPA Alternatives (BPA-Free Products)